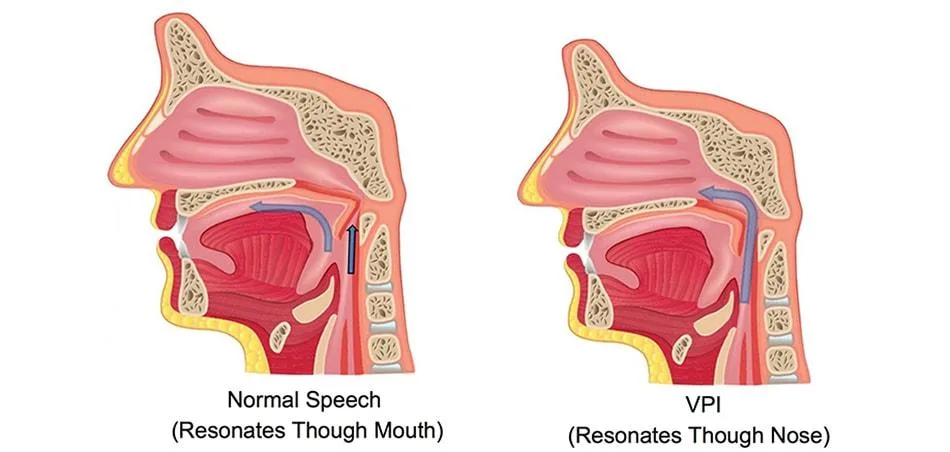
Velopharyngeal Insufficiency (VI) is the inability to temporarily close the connection between nasopharynx and oropharynx, due to an anatomical dysfunction in soft palate (velum), lateral pharyngeal wall or posterior pharyngeal wall. This can be due to insufficient tissue to accomplish closure, or due to some kind of mechanical interference with closure.
An effective sealing between nasopharynx and oropharynx is needed for proper speech, swallowing and breathing. This is accomplished by elevation of the velum and approximation of the lateral and posterior pharyngeal walls during speech and swallowing.
Etiology
- Craniofacial anomalies: Most commonly velopharyngeal insufficiency is seen in children with craniofacial anomalies, especially with cleft palate, either before or after the repair. Despite successful repair, post-repair prevalence of VPI is reported to be 20-50%.
- Submucosal cleft palate: In this situation, no overt cleft is seen, but a failure of the midline fusion of the velar muscles is present often manifesting as a bifid uvula, midline notching of the hard palate, or a bluish line of a visible diastasis.
- Occult submucosal cleft: Characterized by absence or deficiency of the musculus uvulae with a diastasis of the levator veli palatini but without the presence of a bifid uvula or grooving of the oral surface of the soft palate.
On endoscopic examination of the nasopharynx, there is loss of the usual midline convexity of the superior surface of the soft palate with either flattening or a midline groove, consistent with the absence of musculus uvulae. This is sometimes known as the ‘seagull sign’. - Tonsillar hypertrophy: Can cause mechanical interference with velopharyngeal closure.
- Post-surgical
- Following adenoidectomy with or without tonsillectomy – This is a relatively rare condition with a reported incidence of between 1:1,500 – 10,000. Most commonly this happens in children with submucosal cleft. The condition is usually a temporary one, resolving in around 6 weeks time. Permanent VPI after adenoidectomy requiring intervention is reported to occur in approximately 1:1500 adenoidectomies.
- Following orthognathic maxillary advancement surgeries for craniofacial anomalies.
- Following palatopharyngoplasty for treatment of obstructive sleep apnea.
- Post-traumatic
- Syndromic children
- Velocardiofacial / Shprintzen / DiGeorge syndrome / Microdeletions in the long arm of chromosome 22 (22q12) – This is the most common syndromic association of VPI, characterized by overt/submucous/occult submucous cleft palate, conotruncal heart anomalies, mental disabilities, and a characteristic facial dysmorphism.
- Down’s syndrome / Trisomy 21 – This is due to hypotonia of the palatal muscles. But, most of these children most often have a narrow velopharynx and a shallower skull base, resulting in less distance for the palate to traverse to effect closure, this risk is somewhat balanced.
- Kabuki syndrome – Including cleft palate, cardiac abnormalities (mostly coarctation of the aorta), muscular hypotonia, and characteristic facial dysmorphism similar to velocardiofacial syndrome.
- Pierre Robin sequence
- Hemifacial microsomia
- Idiopathic: In 30% of cases the reason for VPI is unknown.
Clinical features
Speech: All phonemes in the English language except the nasal ones (/m/, /n/, /ng/) are produced with oral airflow, for which velopharynx should be closed. The nasal phonemes are produced with nasal resonance, requiring that the velopharynx be open during their production.
Patients with velopharyngeal insufficiency have characteristic hypernasal resonant voice associated with nasal emission of air (escape of air through the nasal passage with speech), nasal turbulence, and inability to produce oral phonemes/ plosives. Parents often complaints this as unintelligible speech.
Children may raise their voice to overcome the nasal escape which results in vocal strain and formation of vocal cord nodules. Such children may have hoarseness of voice also.
Nasal regurgitation of feeds: Severe velopharyngeal insufficiency results in regurgitation of solid foods and fluids through the nose especially in infants.
Recurrent Sinusitis / Middle ear diseases: Persistent nasopharyngeal reflux in older children and adults can cause recurrent or persistent sinus inflammation (sinusitis), ear infections or middle ear effusions.
Diagnosis
History: A detailed history involving developmental milestones, past medical and surgical treatments will be helpful in syndromic children. History of any hearing loss, recurrent sinus infections should also be collected in younger children.
Physical examination: A thorough examination of the oral cavity is needed. Look for any obvious palatal clefting. Palpation of the midline of the soft palate may reveal an occult submucous cleft.
The child should also be evaluated for any syndromic features, craniofacial dysmorphism, and presence of cardiac anomalies.
Speech Analysis: Simple listening and assessment of the child’s speech by a speech pathologist can make the diagnosis and is considered as the gold standard of diagnosis. There will be hypernasality of the voice.
The most commonly used perceptual sound evaluation scales is McWilliams and Phillips / Pittsburgh Weighted Speech Scale. This weighted scale rates five components of speech including nasal emission, facial grimace, nasality, phonation, and articulation. Points are assigned for each subgroup and summed to give an overall score that can be used to track outcomes.
In a large retrospective cohort, assessment of perceptual speech features such as hypernasality and nasal rustle was useful in predicting relative velopharyngeal gap size in the absence of instrumental assessment.
Nasometry/Nasalance: is an objective technique for assessment of nasality of speech, by measuring the ratio of sound output from nose compared with sound output from the mouth for a given spoken word or phrase, expressed in percentage. The score is then compared to normative data, such as with the MacKay-Kummer Simplified Nasometric Assessment Procedures (SNAP) test. A score 3 standard deviations above the mean indicates hypernasal resonance, i.e., speaking with too much airflow and resonance in the nasal cavity.
Flexible Nasapharyngo-laryngoscopy (NPL): NPL scopy provides an excellent view of the nasal surface of the palate and may provide the only sign of a submucous cleft palate. NPL helps in visualizing the velopharynx during the speech, quantify the degree of mobility of the lateral pharyngeal walls and velum, assess the orientation of the levator veli palatini musculature, and estimate the gap size in order to plan for appropriate surgical management.
NPL Scopy will also allow examination of the larynx and helps in the diagnosis of an associated vocal cord lesion as explained above.
To avoid the variations in NPL scopy reports, the Golding-Kushner scale can be used which is one subjective numeric scoring system used to evaluate the multidimensional mobility of the velopharyngeal port.
Multiview Video Fluoroscopy: This procedure is performed as a collaborative effort between a radiologist and an SLP.
This is a radiological evaluation in which a small amount of high-density barium is instilled into the child’s nose, to coat the nasopharynx. Traditionally three Xray views are then obtained while the patient articulates phonemes that require increased oral airflow.
- The anteroposterior/frontal view helps in the assessment of lateral pharyngeal wall motion.
- The lateral view allows visualization of posterior pharyngeal wall, and tongue.
- The base view allows assessment of the sphincter closure pattern.
The advantage of the test is that children who are non-compliant with NPL can better tolerate this. Exposing to ionized barium, X-ray radiations and positioning difficulties are the potential limitations of this study.
Magnetic Resonance Imaging (MRI): The advantage with MRI is that it is noninvasive and there is no risk of exposure to radiations. Many studies have shown that MRI is a better diagnostic tool than videofluoroscopy for visualizing the anatomy of the velopharynx.
The long-time needed for acquiring images, chances of artifacts due to palatal movements are its drawback. In addition, MRI cannot be done in claustrophobic children. Currently, MRI is considered for an overall evaluation of anatomy in children planning for surgery.
Conservative management
Treatment of velopharyngeal insufficiency should begin as soon as possible to promote good speech habits. As a young child develops speech, they develop lifelong speech habits. The persistence of poor speech habits is often hard to change later.
The treatment options in VPI depends on various factors like etiology, age at presentation, length of time of VPI, presence or absence of articulatory compensations, etc. The available treatment options are speech and language therapy, prosthetic obturator or palatal lift appliances and surgical correction (Pharyngoplasty).
Speech / Resonance Therapy
Speech therapy is useful when the insufficiency is a small one, when there are associated articulation errors and in postoperative patients. But as there is a specific anatomic deficiency, the role of speech therapy is limited especially in moderate to severe VPI.
Speech therapy is not indicated when: (a) nasal emissions are present in all non-nasal consonants with subsequent hypernasality related to a short or poor moving palate or excessively deep pharynx, (b) nasal air loss related to a palatal fistula, or (c) articulation errors related to severe dental malocclusions that require physical management and (d) in atypical anatomy.
Simple tools (e.g., cold mirror, paper paddle) or commercial devices like See-Scape, Nasometer etc can be used for visual feedback especially in children with associated hearing impairment.
Obturators, Prosthesis and Nasal Valve
Common prosthetic appliances used in the treatment of VPI include palatopharyngeal obturators, palatal lifts, or pharyngeal bulbs.
Obturators and prosthetic devices are indicated in the treatment of VPI when
- there are any contraindications for surgery (e.g. airway obstruction, obstructive sleep apnea, cardiovascular complications)
- when the cause of VPI is neuromuscular in nature
- when there are wide clefts in with lack of available tissue compromising potential repair techniques
- or as a temporary measure until surgery can be performed.
Obturators can be attached to the palate or teeth and can substitute for tissue deficiency.
Palatal lifts are used when the dynamic motion of the palate is poor due to neuromuscular etiologies. Palatal lifts reduce the distance the palate must traverse for producing adequate closure.
In general, prosthetic management of VPI is not generally a first-line approach due to compliance issues and the need for adequate stable dentition.
A one-way nasal valve fitted on either nostril allows the patient to breathe in through the nose but stops nasal airflow with exhalation or speech.
Surgeries for VPI
Most children with VPI need surgery to prevent air from escaping through their nose.
Surgical correction is indicated when there is evidence of anterior insertion of levator muscles, in cases of submucous cleft or cases were palate repair in the form of Z plasty is unsuccessful.
The most common types of surgery for VPI are Furlow palatoplasty, sphincter pharyngoplasty, posterior pharyngeal flap, or a posterior pharyngeal wall injection augmentation. Selection of surgical procedure depends on the mobility of the lateral pharyngeal walls, the degree of velar elevation, and the size of the defect.
Posterior pharyngeal flap
First described by Schoenborn in 1875, the goal of a posterior pharyngeal myomucosal flap is to effectively obturate the nasopharynx with a biologic obturator.
A superiorily based flap (with base is at the level of the arch of C1) is raised from the posterior pharyngeal wall and is secured to the midline of the nasal surface of the soft palate. The flap forms a static bridge with lateral ports on either side that are closed by medial movement of lateral pharyngeal walls.
This is the preferred method in children with good lateral pharyngeal wall motion, and who have a persistent central gap due to poor palatal motion, as is common following repair of a cleft palate.
Disadvantages:
- As the flap is entirely static it relies on lateral pharyngeal wall movement to effect closure. So only children with some degree of lateral wall motion will get benefited from the procedure.
- Shrinkage of the flap can happen, which can be prevented by the lining of a flap with as much nasal mucosa as possible.
- Too wide flap can narrow the lateral ports resulting in over-correction and hyponasal speech.
- Postoperative airway obstruction can occur, usually within the first 24 hours and in one series resolved within 2 days in more than 90% of patients.
Sphincter pharyngoplasty
This technique mainly employs medial transposition of flaps from the lateral pharyngeal walls. Mainly involve palatopharyngeus muscle and hence the success of this procedure depends on active contraction of the transposed muscle.
Hynes first described pharyngoplasty in 1950 as the elevation of two superiorly based flaps comprising the right and left salpingopharyngeus muscles and overlying mucosa. The flaps are then inserted as high as possible in the posterior pharyngeal wall at the projected level of contact with velum. Hynes pharyngoplasty results are associated with better nasalance score.
Orticochoea modified Hynes technique so that the superiorly based flaps involve the posterior tonsillar pillars with the underlying palatopharyngeus muscles too. In addition, two small inferiorly based posterior pharyngeal wall flaps are also elevated. The lateral flaps are then sutured with the posterior wall flap resulting in a lower down insertion below the projected point of contact with velum.
Jackson modified Ortichchoea technique by sewing the lateral flaps onto a superiorly based posterior pharyngeal wall flap, allowing higher placement of the lateral wall flaps.
A success rate (correction of hypernasality) of 78-100% are reported with pharyngoplasty. Postoperative incidence of hyponasality is reported to be 12-17%.
Posterior pharyngeal wall augmentation
Usually indicated in children with a mild to moderate degree of VPI measuring at most 1-3 mm and a clearly evident small velopharyngeal gap.
A superiorly based posterior pharyngeal wall flap is elevated and rolled it onto itself creating a mound of tissue which ultimately scars and provides for a contact point in the soft palate.
Posterior pharyngeal wall augmentation can also be done by using various materials like cartilage, fat, fascia, paraffin, silicone, acellular dermis, polytetrafluoroethylene, and calcium hydroxylapatite. Advantage of injection methods is the ability to precisely locate and to endoscopically target only the area of deficiency. Problems with extrusion (highest with silicone), migration, resorption (fat, collage), and infection have been reported.
Palatoplasty
Children with a submucous cleft palate or congenitally short palate and associated VPI may benefit from a palatal lengthening procedure such as a Furlow palatoplasty or V-Y pushback palatoplasty.
The V-Y method uses soft tissue elevated from the hard palate whereas Furlow method involves using opposing Z-plasties.
Tonsillectomy
Tonsillectomy can be curative when velopharyngeal closure is obstructed secondary to hypertrophic tonsils.
Complications of surgery
Airway obstruction: Usually reported within the first 24 hours. Placement of nasal trumpets overnight is helpful in the management of airway obstruction. Because of fear of airway obstruction, patients are usually admitted for overnight and discharged on the next day.
Bleeding: Moderate to severe bleeding requiring transfusion is generally rare after sphincter pharyngoplasty or pharyngeal flap surgeries. Presence of aberrant/medialized carotid arteries is a risk factor for catastrophic bleeding children with velocardiofacial syndrome.
Obstructive sleep apnea: The reported prevalence of OSA following surgical correction of VPI varies widely from 4-90% and is usually seen after pharyngeal flap surgeries. But the severity decreases with time so that by 12 months post-op significant OSA is rare irrespective of the type of pharyngoplasty. In the case of severe persistent OSA after posterior pharyngeal flap, division of the flap generally results in the improved nighttime airway and persistent speech benefit.
Careful preoperative airway evaluation and consideration of preoperative adenotonsillectomy 2 months prior may be useful in preventing such untoward outcomes.
As stated above injection augmentation of the posterior pharyngeal wall is associated with problems like extrusion, migration, resorption, and infections.
Outcome and Prognosis
Rogers et al in a retrospective study involving 19 patients with VPI, found that 18 of whom had a history of cleft palate, had significant speech improvement following surgery using a wide, superiorly based pharyngeal flap.
A literature review by Wagner et al in 2017 for VPI associated 22q11.2 deletion syndrome found that treatment with a posterior pharyngeal flap resulted in successful outcomes in 85% of patients.
Setabutr et al indicated that in patients who undergo pharyngeal flap surgery, older age at the time of surgery is a risk factor for revision. The revision rate was 20% (12 patients), with age being the only covariate found that significantly correlated with a greater chance of revision surgery.
Barr et al validated a new disease-specific instrument, the Velopharyngeal Insufficiency Quality of Life (VPIQL) survey, by using it to survey caregivers of children with VPI in conjunction with another generic pediatric quality-of-life instrument. This study identified that children with VPI and their parents perceive reduced quality-of-life when compared with age-matched healthy controls.
Boseley et al in their prospective cohort study implemented the Pediatric Voice Outcome Survey (PVOS), an instrument used to assess general voice-related quality-of-life and showed that parents perceived at least short-term improvements in functional outcomes and quality-of-life following surgery for VPI in their children.
Prevention of Iatrogenic VPI
A number of preoperative predictors are reported in the literature which can alert the surgeon regarding the possibility of post-adenoidectomy VPI. These include the presence of submucous cleft palate, anterior dimpling of the soft palate, a history of nasal regurgitation of fluids, neurological disorders, pre-existing hypernasality of speech or a family history of cleft palate or VPI. Witzel et al. found that over 30% of patients with post-adenoidectomy VPI had such identifiable risk factors. Children with facies consistent with the velocardiofacial syndrome are also at risk of post-surgical VPI.
References
- Wagner RD, Wolfswinkel EM, Buchanan EP, Khechoyan DY. Surgical Outcomes for Speech Surgery in 22q11. 2 Deletion Syndrome: The Dilemma of Persistent Velopharyngeal Insufficiency After Pharyngeal Flap Operation. Journal of Craniofacial Surgery. 2017 Jul 1;28(5):1320-4.
- Rogers C, Konofaos P, Wallace RD. Superiorly Based Pharyngeal Flap for the Surgical Treatment of Velopharyngeal Insufficiency and Speech Outcomes. Journal of Craniofacial Surgery. 2016 Oct 1;27(7):1746-9.
- Setabutr D, Roth CT, Nolen DD, Cervenka B, Sykes JM, Senders CW, Tollefson TT. Revision rates and speech outcomes following pharyngeal flap surgery for velopharyngeal insufficiency. Jama facial plastic surgery. 2015 May 1;17(3):197-201.
- Shprintzen RJ, Marrinan E. Velopharyngeal insufficiency: diagnosis and management. Current opinion in otolaryngology & head and neck surgery. 2009 Aug;17(4):302.
- Dudas JR, Deleyiannis FW, Ford MD, Jiang S, Losee JE. Diagnosis and treatment of velopharyngeal insufficiency: clinical utility of speech evaluation and videofluoroscopy. Annals of plastic surgery. 2006 May 1;56(5):511-7.
- Woo AS. Velopharyngeal dysfunction. InSeminars in plastic surgery 2012 Nov (Vol. 26, No. 04, pp. 170-177). Thieme Medical Publishers.
- Rudnick EF, Sie KC. Velopharyngeal insufficiency: current concepts in diagnosis and management. Current opinion in otolaryngology & head and neck surgery. 2008 Dec 1;16(6):530-5.
- Barr L, Thibeault SL, Muntz H, et al. Quality of life in children with velopharyngeal insufficiency. Arch Otolaryngol Head Neck Surg 2007; 133:224–229.
- Boseley ME, Hartnick CJ. Assessing the outcome of surgery to correct velopharyngeal insufficiency with the pediatric voice outcomes survey. Int J Pediatr Otorhinolaryngol 2004; 68:1429–1433.